Vivodyne has come up with a way to grow complex combinations of human cells in the lab to more realistically test how drugs are going to affect you.

Vivodyne CEO Andrei Georgescu (L) and Chief Commercial Officer Susan Billings (R).
VivoDyne
Only about 10% of drugs in development ever make it to market. That number has remained largely unchanged since the early 1990s, despite milestone advances in science — genomics, automated systems for conducting experiments and artificial intelligence. One big reason: lab research models that can’t accurately capture how drugs will behave in humans. That’s something that needs to change as multiple failures are a big reason why it can cost more than $2 billion to develop a drug.
“These AI models are not creating drugs that are substantially better than human designed drugs,” Andrei Georgescu, CEO of Vivodyne told Forbes. There’s just not enough lab data for models to work with, he explains. That’s because before a drug goes to human trials, it’s mostly tested either on single cells or animals, which gives scientists a good idea of how they work in those contexts, but not how they work in a patient. “The data that we have to build these drugs on and validate these drugs with is simply not translatable to the complexity of humans.”
That’s where Georgescu’s company, Vivodyne, comes in. It’s building human organ tissues in the lab that can be used for preclinical testing of potential drugs, as well as AI systems that can rapidly collect and analyze data from those tests. It’s a replacement, he said, for the large-scale, single-cell lab models or animal testing that dominate the market today, which enable large-scale screening for treating infectious diseases or certain cancers, but they’re also labor intensive and not as useful for more complex diseases. On Wednesday, the company announced that it had raised a $38-million seed round led by Khosla Ventures. Bison Ventures, MBX Ventures, Kairos Ventures and CS Ventures also participated.
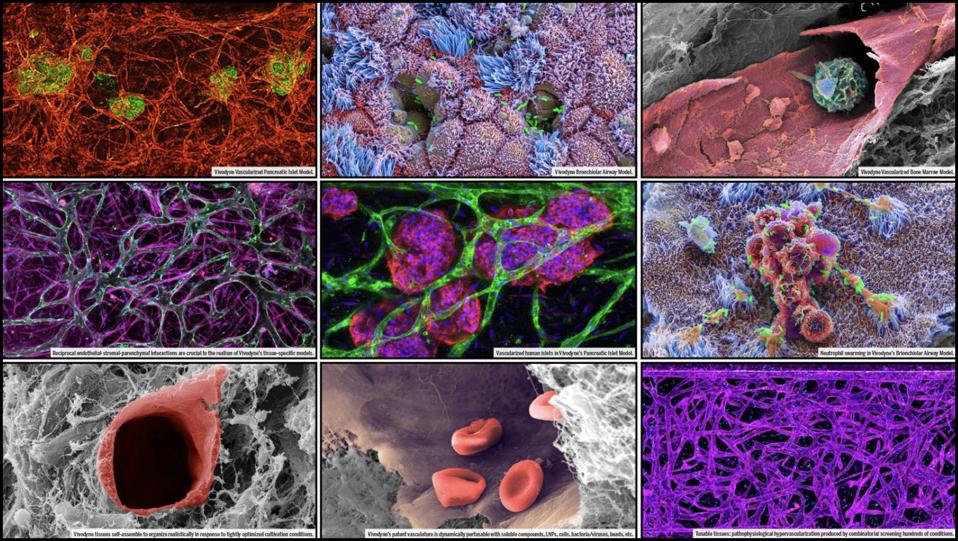
Microscopic images of different types of human tissues grown by Vivodyne.
Vivodyne
Vivodyne’s technology was borne out of Georgescu’s research while working in the lab of Dan Huh, 49, at the University of Pennsylvania. Huh was one of the key researchers behind the first “organ on a chip”, a technology which has enabled scientists to build small scale models of biological systems for lab research. The two founded Vivodyne in the spring of 2021 in a “garage just off-campus from Penn,” Georgescu, 33, says.
Vivodyne’s key technological innovation, Georgescu says, is the ability to grow 22 different types of human tissue that can be used to model blood flow and other interactions between different parts of the body, rather than a single cell. What’s more, he says, Vivodyne’s systems don’t just grow normal red blood cells, but also white blood cells and other cells that enable it to simulate immune responses, which gives researchers a better way to predict how they’ll behave in people.
In addition to growing models of organ systems, Vivodyne can also introduce those organs to different diseases, so that it’s possible to simulate how a drug might treat that disease. For example, Vivodyne can grow lung tissue, infect it with a respiratory disease, and then subject it to various treatments, cataloging the results. “So we’re able to generate clinical human data before clinical trials and before testing on real people,” he said.
What makes Georgescu’s tissue model particularly promising is its scalability, which far exceeds current processes for growing tissue for academic research. There are “tens of thousands of these tissues we can grow at once,” he said, “with reproducibility and extremely tight control thanks to the full robotic automation of the process.”
Alex Morgan, a partner at Khosla Ventures, says it is precisely this scalability that most excites him about this investment. Not only does it hold promise for improving discovery of drugs, he told Forbes, it also provides useful data that machine-learning systems can use to help biotech companies design better drugs. “There’s so much data being collected” that can be interpreted by AI systems, he said, that it will help Vivodyne be a huge driver for its customers.
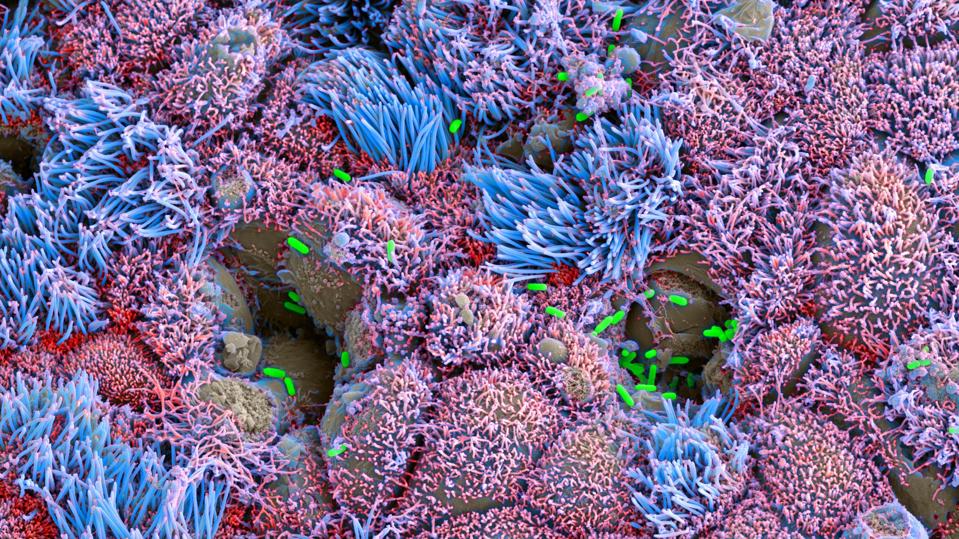
A colorized microscopic image of lab-grown tissue infected by bacteria (green).
Vivodyne
Speaking of those customers, Georgescu says Vivodyne is already working with several companies to assist in preclinical drug development and to troubleshoot disappointing results in human trials. With the new infusion of capital in this seed round, he told Forbes that Vivodyne will be able to develop more tissue systems for its platform, scale up its automation and build up its base of talent.
The overarching goal for his company, Georgescu says, is to turn it into the biological equivalent of a “large-scale data center” that provides scientists with unprecedented capabilities for researching human disease. “That is the future we’re working towards.”
This article was first published on forbes.com and all figures are in USD.


