Tetratherix built a $255 million ASX company around a single injectable polymer. Now that same material is being used in everything from dental surgery to prostate cancer treatment and GLP-1 delivery.

Tetratherix grabbed the market’s attention in March when it announced its patented polymer was going to be used for the slow release of GLP-1 inhibitors, peptides and hormones.
Its share price surged on its linkage to these fashionable molecules, fell and is surging again after the $3 million-a-year deal with Superpower Health Inc was announced on March 16. A week earlier the market hadn’t budged on good news about Tetratherix’s clinical trials into using the exact same compound as a spacer in prostate cancer treatment.
It had moved a lot though when the company announced in January that the same compound was going to be distributed by global dental tech giant Henry Schien for use in dental surgery.
And the same compound is heading towards FDA approval this year in bone regeneration.
The $255 million valuation is built entirely on this Swiss Army knife of a polymer, essentially a plastic, that spun out of the University of Sydney in 2014.
Understanding how it came to be, and the path that led Ali Fathi and Terence Abrams from a university lab to an ASX‑listed company, is key to seeing why investors, clinicians and potential partners are paying close attention today.
Tetratherix began with Ali Fathi’s PhD at the University of Sydney. The chemical engineering student from Iran started with the idea of making cartilage in the lab, then inserting it into such needy places as people’s knees. Until he saw such surgeries being performed.
“I realised that this is a joke,” Fathi tells Forbes Australia. “It’s not gonna be commercialised. They did so much damage to the surrounding tissue.”
The then 22-year-old who’d been in Australia four years didn’t want to waste his time doing something that wasn’t going to be commercial. So he decided to devote his PhD to finding a polymer that could be injected into the body to help the body repair its own cartilage or bone.
He had four criteria. It needed to: be injectable; cause zero inflammation; stay where it was put; and break down and leave the body over time.
Within the first year, 2011, he had combined four monomers that had such characteristics, and baked them into a single polymer that they now call Tetramatrix and which is the entire backbone of Tetratherix’s $255 million market capitalisation.
But things weren’t nearly so clear 15 years ago.
“By 2011 we had something patentable, but we didn’t know if it’s going to kill animals, whether it’s biocompatible, none of that. We just knew that we had a new plastic,” says Fathi.
Animal studies followed, and by 2014 he had his PhD, and an injectable polymer that appeared safe and helped animals heal bones.
“I’m coming from a family that is very much business oriented, so I knew I wanted to make a company out of this. My dad is in the oil and gas industry in Switzerland, so I had that background … it just gears you towards that natural progression.”
He took his polymer to San Francisco to pitch Google Ventures. He got a “yes”, he says, but had to move to the US to get the money, so he gave them a “no”.
Back in Sydney, he teamed up with Terence Abrams, who was finishing his chemical engineering undergrad degree. They got the patents out of the uni, formed a company and found two seed investors.

Within 18 months they had completed human trials.
“The first trial was 10 patients,” says Fathi. “When you have a tooth extraction, you have a gap in the bone. So think about it as a cup. We injected the product into this cup … and we followed up after three months. We showed that the gum heals better.”
They’d chosen dentistry because clinical trials in mouths cost about $10,000 per patient. In knees they cost $50,000 to $60,000, Fathi says. “It’s a very honest answer. Very important because that’s the culture of the company. That’s the reason we are still here after 10 years.”
They completed four human trials with a payroll of zero.

“We built the lab downstairs,” says Fathi. “We were cleaning the lab. We were making the polymer. We were doing the bookkeeping. It was a two-man shop by design, because we wanted to maximize the R&D strength of the company without getting too diluted by VC funding.”
The other reason they chose dentistry was that after the dentists had drilled screws into the new bone material to put crowns in, they could sample the bone from the screw holes. “We saw bone, we saw soft tissue, we saw that after three months our product was completely gone without causing any damage to the surroundings. So, like, a tick, tick, tick. All we wanted to see.”
The results led to a $1 million grant from the NSW Medical Devices Fund.
At the ceremony marking the awarding of the grant, Fathi got speaking to Will Knox, who’d recently successfully exited a business he’d founded, uHealth, which had developed a crustacean shell derivative to regenerate cartilage.
When they told him they had a material that was completely bio-inert and caused no rejection from the body, he didn’t believe them. It was the holy grail of regenerative implants. And here were these two guys telling him they’d built a lab and a manufacturing facility that had full ISO [International Organization for Standardization] certification.

“Two blokes sitting in some warehouse in Alexandria having a full quality system that’s certified by the world’s biggest certifying body,” recalls Knox. “There’s no fucking way. That’s a huge deal in our game.”
But he dropped in on their lab/office/factory a few days later. Just in case it was true. He still finds it hard to believe. “They’d designed the whole quality management system, the paperwork, the certification, built downstairs with their own hands, and got it certified from 2017 onwards as a startup with two blokes.”
Fathi says there were a lot of late nights. “Many startups don’t do the quality system because it’s not a fun part of the job. It’s annoying, but it wasn’t a lifestyle for us … To be able to make a business out of research, you need a quality management system.”
Knox knew that most start ups did quality systems at the end, when they have large teams. “These guys had been doing that from day one because they’d always had this mindset of this is going to be a generational Aussie MedTech that can be the next Cochlear. That’s what drew me into the opportunity.”
Like chewing gum
Fathi squeezes out some Tetramatrix onto his hand. It goes from being like a clear silicon gel, to a thick, sticky white glug, the texture of chewing gum, as soon as it warms to his body temperature.
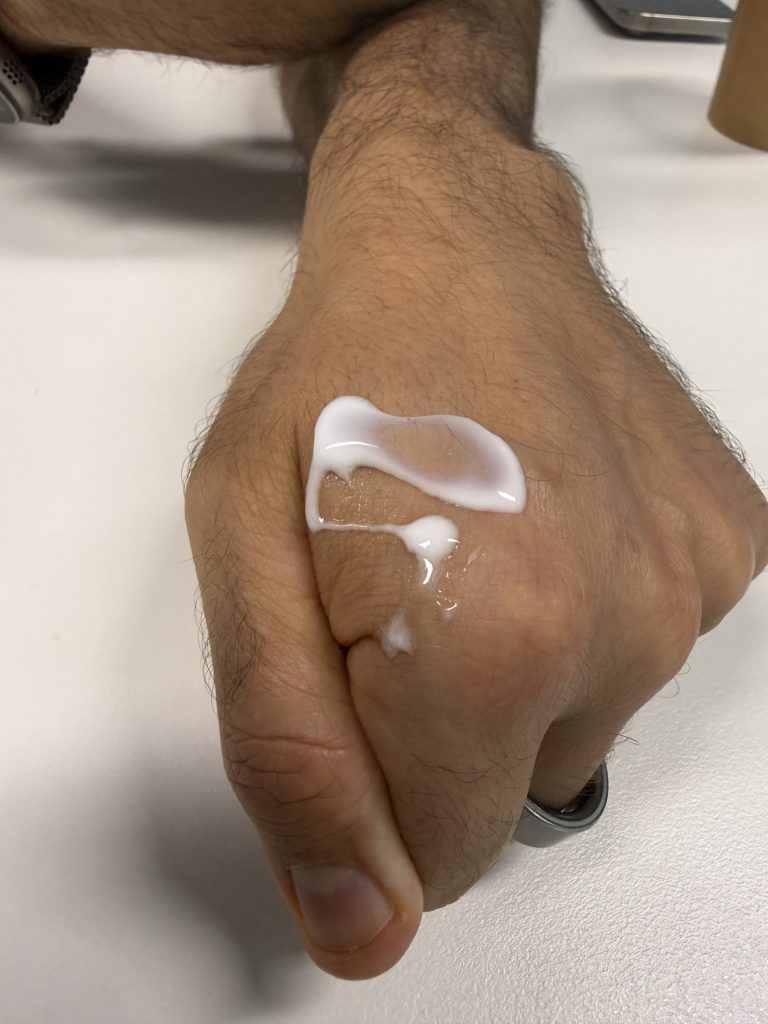
When Knox saw this Spakfilla for humans, his mind went straight to its multiple uses. He was looking at a “platform technology”. The uses have come to include:
- In dentistry, where it has been shown to heal bone and soft tissue more quickly.
- To regenerate bones by, say, being injected into a shattered wrist with bone granules mixed in to hold all the pieces together while the bone heals itself.
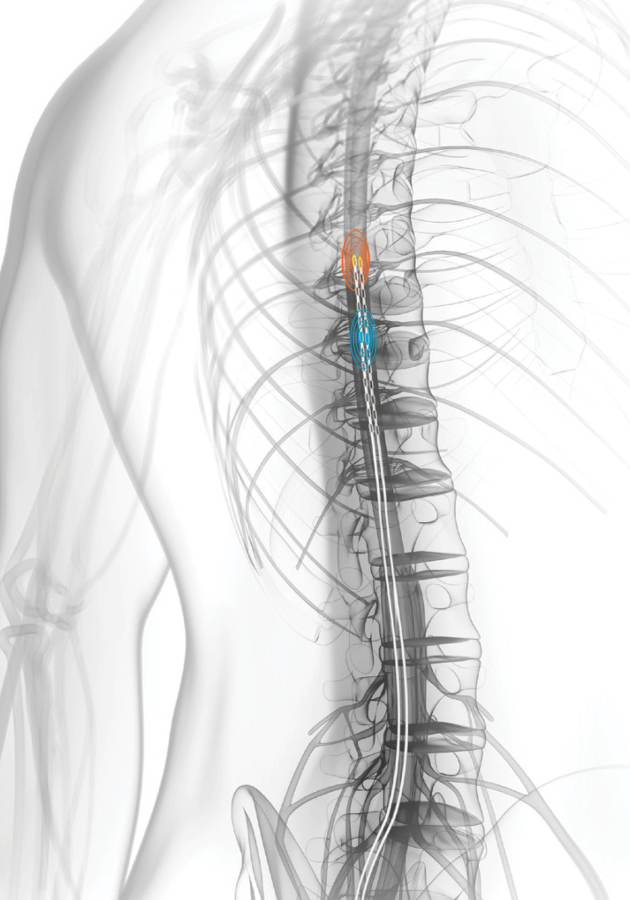
- Cancer treatment, where it could be injected into the space between the prostate and the rectum so there was less chance of damaging other tissue with radiation.
- Healing scars faster after surgery by laying down a slither of it in the wound before stitching. In the world of post-GLP1 surgery to remove flabby skin, there was going to be miles of suturing that needed seamless healing.
- Slow-release drug delivery. By infusing it with a drug, as the Tetramatrix slowly breaks down in the body, the drug would be released. This is the use that got the market excited in March about GLP-1s.
“It’s the same technology, but it’s got all these different applications,” says Knox.

“I’d come from the Australian medtech sphere, which had always had this phrase of ‘the next Cochlear’ or ‘the next Resmed’ bandied around, but no one’s managed to live up to that for many decades. I’d always wanted that to be part of my career.”
At first, he just put his money where his dreams were. “Then, in 2021, after I’d seen that these guys can actually deliver what they said they could, and more, I came on as CEO,” says Knox.
They could see the finish line. But how to get there? “We now needed the funds to execute,” says Knox.
Sydney-based Ryder Capital had invested in 2020. Xero founder Rod Drury came in next with his Radar Ventures.
Drury liked that the combination of founders. “You’ve got the crazy-bad scientist guy and a really good commercial person,” Drury told Forbes Australia. “What I also liked is that they were very curious, talking to me around how you build platforms, and they saw the parallel between the platform we created at Xero and what they were doing with their technology, which is pretty horizontal, and then they can put a bunch of verticals over.
“That led to the idea of maybe we should do an early IPO like Xero. And I liked that they listened to the ideas and they act on them really quickly.”
Because, as Knox looked at his schedule for the first five potential uses of Tetramatrix, he wasn’t sure venture capital could keep up.
“The cadence with which we develop product and grow the business is so fast that trying to have a VC or a private funding round, particularly in our space here in Australia, and keep up with that cadence was just going to slow us down.”
They decided to go with faster access to capital by listing.
“And we don’t want to create a business that is going to be bought by a big multinational while we ride off into the sunset.”

They had a pre-IPO fundraising round in 2024 that was quickly oversubscribed despite it being the worst year for IPOs since the GFC.
“We thought, ‘We’ve got something here, so let’s pull the trigger and get it done,’” says Knox.
They’d only spent “$12 million to $13 million” to get to the IPO and after it, the three founders still hold about 40% of the company. Fathi has 28%, Knox 7%, and Abrams 5% while Drury’s Radar Ventures has 13.3%.
They raised $25 million on the June 2025 Initial Price Offer of $2.88 a share. It has run as high as $5.80, but has never sunk below its IPO price.
The next problem was how to get all those uses to market. Knox wanted to keep all manufacturing in Australia, but to team up with the global market leaders in each field to get it out to the world.

Validation of that idea came in January 2026 when the company announced it had signed a deal with Nasdaq-listed Henry Schein Inc to distribute Tetramatrix technology globally for dental uses.
“They’re the world’s biggest provider of dental products,” says Knox. “They’ve recognised the appeal of this for the global application in dental surgery … which is really exciting for us as a small Aussie business that’s come out of Sydney Uni.
“We’ve got two or three of the biggest orthopaedic companies in the world who have approached us about working with them in a similar way … So it’s exciting.”
Animal studies are underway to use Tetramatrix as a spacer in eye surgery. “We’ve been approached by Alcon, which is again the number one player in eye procedures and ophthalmic technologies around the world,” says Knox.
And then they announced the deal with Superpower Health in March with the US company paying US$3 million per year for up to 10 years to use it in its slow-release drug capacity. “Superpower will purchase Tetramatrix platform polymer (branded as STEPP) for nasal delivery of longevity and metabolic compounds such as GLP-1, peptides and hormones in the US market,” the release said.
Using the same Tetramatrix polymer, the technology forms a sticky layer on the nasal lining, helping drugs remain in place for absorption while protecting fragile molecules from degradation.
While the STEPP delivery mechanism was years away from any FDA approval, they are “pushing a novel pathway by using compounding pharmacy pathways to collect dynamic, real world evidence at scale,” a company spokesman told Forbes Australia in a statement. “This will mean they’ll generate real world consumer demand as well as safety and efficacy data from thousands of patients that we will start seeing in months, not years.”

Knox explains something of the strategy. “Rather than trying to create a product and then shoehorn it into the market, what we do with all these applications is before we even pursue the proper design development process, we go to the end user and say, ‘If this is going to be used for this application, how much would it cost? How much does it need to be reimbursed for? Can we make it cheap enough? And can we make it at scale?’ If any of those answers are ‘no’, then we just don’t pursue it because we don’t want to end up being another science experiment.”
The use for healing scars is in clinical trials and has proven safe.
The dental and orthopaedic trials are complete and are going to be submitted to the US FDA in April, seeking final approval to go to market.

While Tetratherix manufactures at its current Sydney laboratory, it is building a new plant with room to be ten-times larger. “The good thing is, because we’ve done 10 to 15 years of R&D, the sunk cost of capital into the business is almost like a defined line in the sand,” says Knox. “There isn’t a huge amount of R&D we now need to do.
“But what we can now do is manufacture at scale and provide a compounding high gross margin opportunity to investors.”
They are hoping to have the first five products in market by 2029, says Knox. “And we’ve then got a pipeline of applications coming behind those.”
“So it will grow over time. There’s so many other areas that we can get onto. We had a call this morning with a US group that’s got an AI drug-discovery engine that’s got a knee osteoarthritis drug that they want us to help them commercialise because of our polymer technology that can help retain the drug within the knee joint.
“So there’s all these different partnership opportunities.”
The input ingredients are cheap petrochemicals. One of their 10-litre reactors can make 4,500 doses of the dental product in a day.
“We have been approached several times to move the whole business across to the US, whether that’s to source US funding or just to be an American business, but we’ve vehemently said no at all times because we want to create a local ecosystem to help the industry, help young researchers do more of this stuff to export this type of R&D and technical expertise around the world.
“As I said, this is the most genuine opportunity for an Australian business to be ‘the next Cochlear’.”
Want to see more Forbes articles on your feed? Tap here to make Forbes Australia a preferred source on Google.
Look back on the week that was with hand-picked articles from Australia and around the world. Sign up to the Forbes Australia newsletter here or become a member here.